
For the GAPDH strip, blocking was carried out for 2hrs in 5% non-fat dry milk before being incubated overnight at 4☌ in HB9177 at a 1:2000 dilution (0.5µg/ml). Following transfer the membrane was cut between markers into sections with the section containing GAPDH being used for western blot while the remaining sections were incubated in Coomassie dye before being destained in several changes of destaining solution (see HB0739 for recipes). Wet transfer to a PVDF membrane was completed in 90 minutes using 400mA. 
Rat cytosol samples (total protein loading ranging 5-70µg / lane) from were loading onto a 12% acrylamide gel alongside a protein ladder (BioRad Precision Plus dual colour, 1610374) before being run at 60V for 40 minutes followed by 120V for 90 minutes. Method: cytosol fractions were prepared from fresh rat brains following established protocols (Molnar et al., 1993. HB9177 staining strongly correlates (R2 = 0.86) with Coomassie derived total protein staining while showing greater sensitivity. Band intensity was calculated using Image Studio version 5.2.5 (LiCor) and a graph was constructed in GraphPad Prism 9 using a 3-parameter Hill equation curve fit. Detection was accomplished using Clarity Western ECL substrate (BioRad, 1705061) and a Licor Odyssey Fc imaging system (ECL channel: 10 min exposure, 700nm channel: 30 sec exposure). For more detail please see our Western blotting protocol. Following washing the membrane was incubated in secondary antibody (1:10,000 dilution, Polyclonal goat anti-mouse HRP conjugated, Sigma Aldrich A3682) for 2hrs. Each strip was incubated separately with a separate HB9177 concentration with this ranging from 0.8µg/ml (1:1,250 dilution) to 3.125ng/ml (1:320,000 dilution). Strips were blocked for 2hrs in 5% non-fat dry milk before being incubated overnight at 4☌ in HB9177. Following transfer the membrane was cut into strips using Ponceau dye to visualise and cut individual lanes. Rat cytosol samples (20µg / lane) were loaded onto a 12% acrylamide gel alongside a protein ladder (BioRad Precision Plus dual colour, 1610374) before being run at 60V for 30 minutes followed by 120V for 95 minutes. HB9177 shows consistent results with low background at dilutions as low as 1:320,000 (3.125 ng/ml). The membrane was blocked for 2hrs in 5% non-fat dry milk before being incubated overnight at 4☌ in HB9177 at a 1:2000 dilution (0.5µg/ml). Samples (20µg / lane) were loaded onto a 12% acrylamide gel alongside a protein ladder (BioRad Precision Plus dual colour, 1610374) before being run at 60V for 30 minutes followed by 120V for 90 minutes. Other tissue lysates were prepared following established protocols from freshly dissected tissue (see our guide on WB sample preparation). 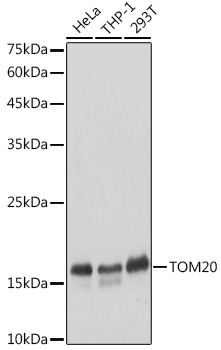
Neuroscience 53:307-326) from freshly collected adult brains. Method: mouse brain and rat brain membrane (P2) and cytosol fractions were prepared following previous work (Molnar et al., 1993. HB9177 revealed a single band of size 34kDa present in all samples with a strong signal to noise ratio.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
